All posts tagged: Clean in Place
Case Study – Food Processing CIP
Problem:
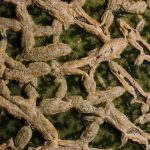
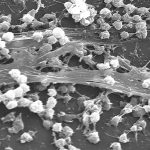
A leading soup manufacturer was repeatedly recording high levels of biological contamination in their CIP process and end-product and was experiencing lengthy batch change-over cycles as a result. Soup stock Quality Control requires that microbe counts are taken after packing which leads to high financial losses if the batch fails and has to be disposed of. The high level of cross-contamination within the existing CIP regime was leading to high batch fail levels and a solution to these losses had to be found.